3D Cell Culturing technology to grow cells so that they retain or recover their in-vivo physiological attributes.
Accelerate to discover
Related topics
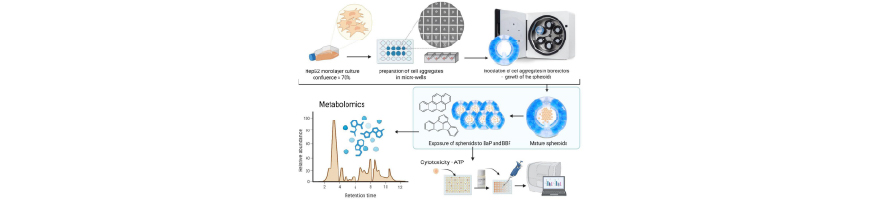
Metabolomic fingerprints of PAH exposure – identifying toxicological biomarkers in dynamic culture
For experimental purposes, 3D cell models (spheroids) were generated by pre-forming cell aggregates using AggreWell™ 400 µm 24-well plates (STEMCELL technologies, Vancouver, Canada) following the manufacturer’s instructions. Cells were seeded at a density of 200.000 cells/well, yielding approximately 170 cells per microwell, in complete growth medium, followed by centrifugation at 100 g for 5 min. The plates with spheroids were incubated overnight at 37 °C in a humidified atmosphere containing 5 % CO2 to allow for cell self-aggregation. Spheroids were subsequently cultured under dynamic conditions using the ClinoStar 2 rotating bioreactor system (CelVivo ApS, Odense, Denmark). To prepare the single-use ClinoReactorsTM (CelVivo ApS, Odense, Denmark), sterile water was added to the humidification beads located in the humidity chamber, allowing them to hydrate at room temperature for 4 hours. Once hydrated, 7mL of HepG2 growth medium was added to each ClinoReactorTM. The units were then placed in ClinoStar 2 system and left to equilibrate overnight at 37 °C in a 5 % CO₂ atmosphere, rotating
at a speed of 12 rpm. On the following day (Day 0), cell aggregates were gently detached from the
AggreWell™ plates by pipetting, inspected under a light microscope to determine their quality, and transferred (approximately 1200 cell aggregates prepared in a single well per ClinoReactor) into the pre-equilibrated ClinoReactor. The bioreactors were then filled with growth medium to a final volume of 10 mL and placed into the ClinoStar 2 system, where they were maintained at 37 °C in a 5 % CO2 atmosphere under continuous rotation (initial rotation speed of 10 rpm). To support optimal spheroid growth, 90 % of the culture medium was refreshed on a 48/48/72-hour schedule. The rotation speed of ClinoReactors was adjusted progressively to accommodate spheroid growth. During each media change, a “gardening” step was performed to remove spheroids that were irregular in size or shape, promoting greater uniformity within the culture. The spheroids were transferred to new equilibrated ClinoReactor units at day 10 to ensure optimal growth conditions, prevent the accumulation of cellular debris, and maintain the integrity of the experimental environment. HepG2 spheroids were cultured over a 21-day period before being exposed to the studied PAH compounds.
Related technologies: 3D cell culturing



